![]() Yamaguchi University School of Medicine
Yamaguchi University School of Medicine
Department of Biochemistry and Molecular Biology
Research Activities
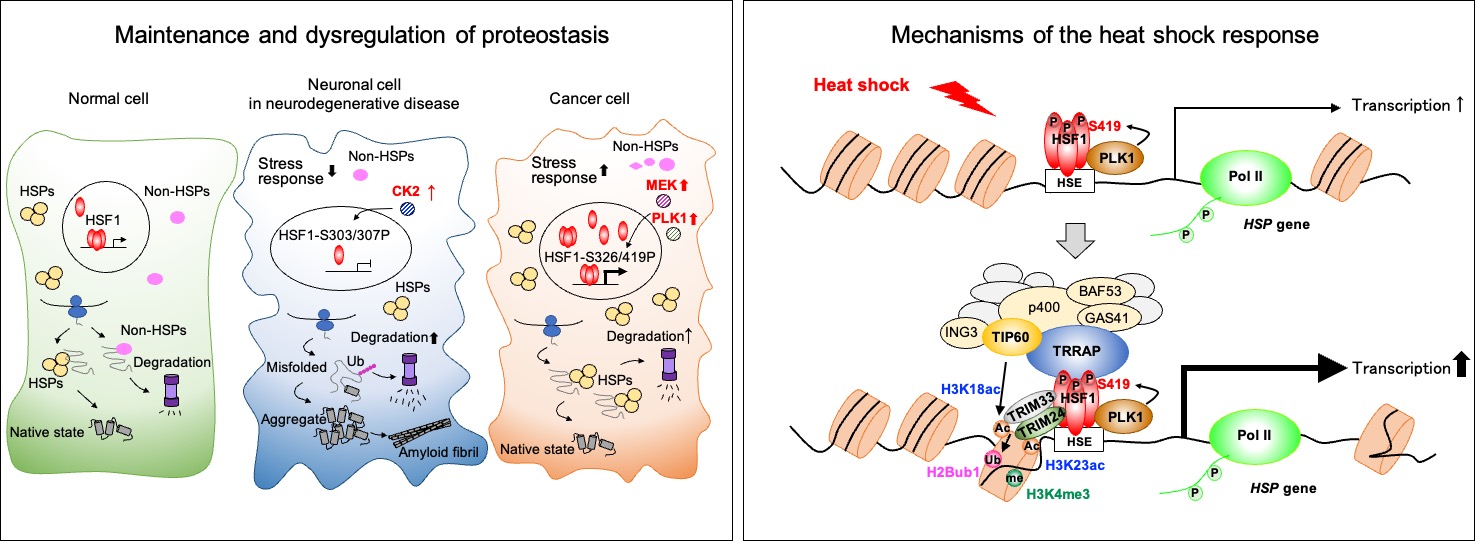
The aim of our research is to understand the molecular mechanisms that regulate proteostasis capacity, with a focus on the heat shock response and heat shock factors (HSFs), and to identify new strategies to ameliorate protein misfolding diseases and cancer.
Proteostasis capacity declines with aging and is associated with age-related protein misfolding diseases, including neurodegenerative disorders. HSF1 and other members of the HSF family play pivotal roles in regulating cellular proteostasis capacity. Accordingly, gain of HSF1 function inhibits the progression of protein misfolding diseases in mice and extends lifespan in C. elegans. Conversely, loss of HSF1 function suppresses cancer progression, as cancer initiation and progression depend on elevated proteostasis capacity mediated by HSF1 activation.
Recently, we have been analyzing the components of the HSF1 transcriptional complex and uncovering signaling pathways that link cellular metabolism to proteostasis capacity. We have already demonstrated that proteostasis capacity is tightly associated with DNA, glucose, lipid, and energy homeostasis within the cell. These studies are expected to identify novel therapeutic targets for patients suffering from protein misfolding diseases and cancer.
Selected Publications
1) Takii R, Fujimoto M, Pandey A, Jaiswal K, Shearwin-Whyatt L, Grutzner F, and Nakai A. HSF1 is required for cellular adaptation to daily temperature fluctuations. Sci. Rep. 14, 21361, 2024, September 12, 2024, DOI: 10.1038/s41598-024-72415-x.
2) Okada M, Fujimoto M, Srivastava P, Pandey A, Takii R, and Nakai A. The Mediator subunit MED12 promotes formation of HSF1 condensates on heat shock response element arrays in heat-shock clles.FEBS Lett. 597 (13), 1702-1717, 2023 (selected as the Editor's Choice)
3) Fujimoto M, Takii R, Matsumoto M, Okada M, Nakayama KI, Nakato R, Fujiki K, Shirahige K, and Nakai A. HSF1 phosphorylation establishes an active chromatin state via the TRRAP-TIP60 complex and promotes tumorigenesis. Nat. Commun. 13, 4355, 2022. DOI: 10.1038/s41467-022-32034-4. (featured in a Nature Communications Editors’ Highlights webpage) (Press release)
4) P. Srivastava, R. Takii, M. Okada, M. Fujimoto, and A. Nakai. MED12 interacts with the heat shock transcription factor HSF1 and recruits CDK8 to promote the heat shock response in mammalian cells. FEBS Lett. 595 (14), 1933-1948, 2021. DOI: 10.1002/1873-3468.14139.
5) A. Katiyar, M. Fujimoto, K. Tan, A. Kurashima, P. Srivastava, M. Okada, R. Takii, and A. Nakai. HSF1 is required for induction of mitochondrial chaperones during the mitochondrial unfolded protein response. FEBS Open Bio 10 (6), 1135-1148, 2020. (selected as the winner of inaugural 2021 FEBS Open Bio Prize)
6) R. Takii, M. Fujimoto, M. Matsumoto, P. Srivastava, A. Katiyar, K.I. Nakayama, and A. Nakai. The pericentromeric protein shugoshin 2 cooperates with HSF1 in heat shock response and RNA Pol II recruitment. EMBO J. 38 (24), e102566, 2019. (selected as a Cover, Introduced in News & Views) (Press release)
7) M. Fujimoto, R. Takii, A. Katiyar, P. Srivastava, and A. Nakai. Poly(ADP-Ribose) Polymerase 1 Promotes the Human Heat Shock Response by Facilitating Heat Shock Transcription Factor 1 Binding to DNA. Mol. Cell. Biol. 38, e00051-18, 2018.
8) M. Fujimoto, R. Takii, E. Takaki, A. Katiyar, R. Nakato, K. Shirahige, and A. Nakai. The HSF1-PARP13-PARP1 complex facilitates DNA repair and promotes mammary tumorigenesis. Nat. Commun. 8, 1638, 2017. (Press release)
9) K. Tan, M. Fujimoto, R. Takii, E. Takaki, N. Hayashida, and A. Nakai. Mitochondrial SSBP1 protects cells from proteotoxic stresses by potentiating stress-induced HSF1 transcriptional activity. Nat. Commun. 6, 6580, 2015. (Press release)
10) R. Takii, M. Fujimoto, K. Tan, E. Takaki, N. Hayashida, R. Nakato, K. Shirahige, and A. Nakai. ATF1 modulates the heat shock response by regulating the stress-inducible HSF1-transcription complex. Mol. Cell. Biol. 35, 11-25, 2015.
11) M. Fujimoto, E. Takaki, R. Takii, K. Tan, R. Prakasam, N. Hayashida, S. Iemura, T. Natsume, and A. Nakai. RPA assists HSF1 access to nucleosomal DNA by recruiting histone chaperone FACT. Mol. Cell 48, 182-194, 2012. (Press release)
Selected Reviews
1) Fujimoto M, Takii R, and Nakai A. Regulation of HSF1 transcriptional complexes under proteotoxic stress. BioEssays 45 (7), e2300036, 2023. (Invited Review)
2)Nakai A. Molecular basis of HSF regulation. Nat. Struct. Mol. Biol. 23, 93-95, 2016 (News and Views)
3) Nakai A ed. 2016. Heat Shock Factor. Springer, Japan. (eBook + Hard Cover)
4) Fujimoto M, Takii R, Hayashida N, Nakai A. Analysis of the heat shock factor complex in mammalian HSP70 promoter. Methods Mol. Biol. 1292, 53-65, 2015.
5) A. Nakai. Novel aspects of heat shock factors. FEBS J. 277: 4111, 2010. (Overview of the Mini-review series)
6) M. Fujimoto and A. Nakai. Novel aspects of heat shock factors: the heat shock factor family and adaptation to proteotoxic stress. FEBS J. 277: 4112-4125, 2010. (FEBS Journal Top-Cited Paper Award)
7) A. Nakai. Heat shock transcription factors and sensory placode development. BMB reports 42: 631-635, 2009.
Message
A cell is the fundamental unit of life. Researchers in the fields of biochemistry and molecular biology seek to understand the fundamental processes within cells that regulate growth, differentiation, cell death, and responses to stimuli. The discovery of new fundamental processes contributes not only to the establishment of novel concepts but also to the understanding and treatment of diseases. Our laboratory welcomes researchers who are enthusiastic about uncovering the truths of nature.
Contact Information
Akira Nakai, MD&PhD, Professor, Department of Biochemistry and Molecular Biology, Yamaguchi University School of Medicine, Minami-Kogushi 1-1-1, Ube 755-8505, Japan. Phone: 0836-22-2214, Fax: 0836-22-2315, E-mail: anakai@yamaguchi-u.ac.jp